Fetal magnetocardiography (fMCG) is an innovative and emerging diagnostic utility aimed at the early identification of fetal heart problems. Its efficacy for diagnosis and management of serious fetal arrhythmia has been acknowledged in the recent American Heart Association Statement on Diagnosis and Treatment of Fetal Cardiac Disease.
In 2016, an experimental fetal MCG system with a just a handful of early QZFM sensors was installed at the University of Wisconsin (Prof. Wakai). Results from the clinical study involving 15 subjects (8 with uncomplicated pregnancies and 7 with pregnancies complicated by fetal arrhythmia or a high risk of fetal arrhythmia) were recently published in JACC Clin. Electrophysiol. (link). The article states, “The main finding of this study is that OPMs can detect fetal rhythm abnormalities with efficacy similar to that of an FDA-approved SQUID magnetometer. Despite the modest number of subjects, we also demonstrated the ability of the OPMs to detect abnormal repolarization, a critical and unique capability of fMCG.”
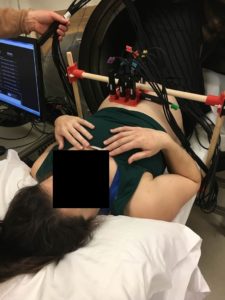
Experimental fMCG setup at U. Wisconsin fMCG (2017) with 9 QZFM sensors and small human-sized (open-ended) magnetic shields.
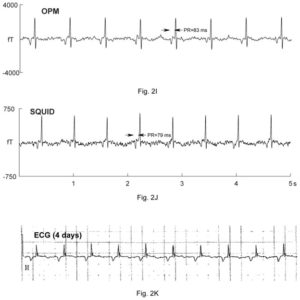
Example data comparing the processed output from the experimental OPM (QZFM) system and from Tristan (FDA approved ) SQUID system. The bottom graph shows postnatal ECG data. The data is from a fetus at 30-1/7 weeks’ with a low atrial rhythm, characterized by a low heart rate, inverted P-wave, and short PR interval]
Dr. Jannet Strasburger (M.D., Medical College of Wisconsin) gives a quick introduction to fetal MCG in a short video presentation below (02:32 min).
Read More: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5841248/